To this add 3-4 drops of the given liquid (or 0.1 g if solid) and warm the test tube on a water bath for about 5 minutes. Now add dilute ammonia solution dropwise until the brown precipitate of silver oxide just dissolves. A brown precipitate of silver oxide forms. Take about 1 ml of silver nitrate solution in a clean test tube and add 2-3 ml of dilute NaOH solution. Presence of aldehydic group is confirmed. 
sodium hydroxide solution.ĭissolve a small quantity of the given compound in a clean test tube and add about 1 ml of Schiff’s reagent and shake it well. Cool and pour the reaction mixture carefully into a beaker containing 15 ml of dil. Heat the test tube for about 1 minute in an oil bath. Take 1 ml of the given liquid in a clean dry test tube (or crystal if solid) and add 1-2 drops of conc. sulphuric acid and shake the test tube to mix the contents.Ī deep blue or green colouration is seen.Īdd distilled water carefully to the test tube.Īdd excess of sodium hydroxide solution to the test tube. Heat the test tube very gently for half a minute and allow it to cool. Take a small quantity of sodium nitrite in a clean dry test tube and add about 1 ml of the given liquid (or crystal if solid). 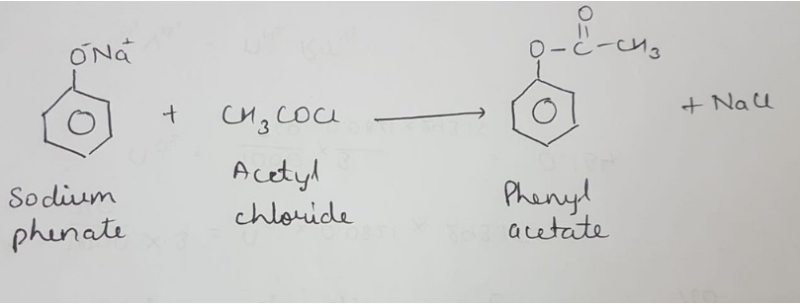
Take about 1 ml of neutral ferric chloride solution in a clean test tube and add 2-3 drops of the given liquid (or crystal is solid). Place a drop of the given liquid (or a crystal if solid) on a moist blue litmus paper. 
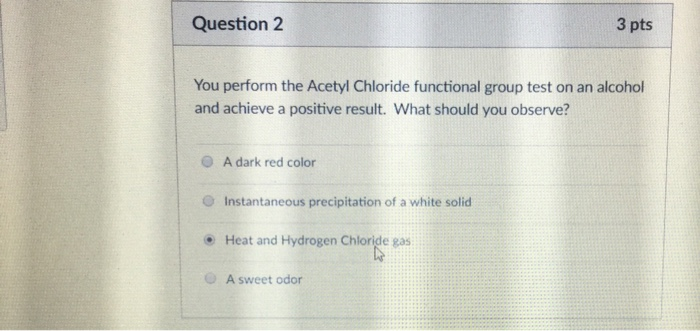
Shake the test tube well and bring a glass rod dipped in ammonium hydroxide solution near the mouth of the test tube.ĭistinguishing Tests for Primary, Secondary and tertiary Alcohols Filter the liquid into another clean dry test tube and to the filtrate, add 2-3 drops of acetyl chloride. Take about 2 ml of the given liquid in a clean dry test tube, add 1 g of anhydrous calcium sulphate and shake well to remove water. Take about 1 ml of the given liquid in a clean dry test tube and add few drops of ceric ammonium nitrate reagent and shake it well. Pour it into about 20 ml of cold water taken in a beaker and smell. Warm the mixture on a water bath for about 10 minutes. Take about 1 ml of the give liquid in a clean dry test tube, add about 1 ml glacial acetic acid and 2-3 drops of conc. Filter the liquid into another clean dry test tube and add a small piece of sodium metal.īrisk effervescence is due to the evolution of hydrogen gas indicating the presence of alcoholic group. Take about 1 ml of the given liquid in a dry test tube, add about 1gram of anhydrous calcium sulphate and shake well to remove water. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
